Abstract
Background: Myeloproliferative neoplasm in blast phase (MPN-BP) is a secondary acute myeloid leukemia (sAML) notable for poor response to chemotherapy and short median survival. In a prior study, we used glycoprotein capture cell surface proteomics to find CD72 as a novel immunotherapy target highly expressed in KMT2A-rearranged B lymphoblastic leukemia, the poorest-prognosis subtype of this disease (Nix et al, Cancer Discov 2021). Given unique driver mutations (i.e. JAK2, CALR, MPL), we hypothesized that the cell surface of MPN-BP would reveal a distinct biological signature and therapeutic targets versus de novo AML. To our knowledge, an MPN BP-specific immunotherapy has yet to be developed. Thus, we took a two-pronged approach: 1) use unbiased proteomics discovery to identify surface proteins enriched in MPN-BP cell lines that have potential for immunotherapy development; and 2) generate immunotherapy against a known target (surface CALR) with specificity to a subset of MPN-BP.
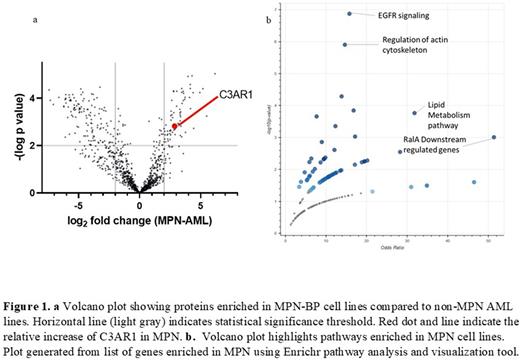
Methods and Results: Toward the first strategy, we profiled the cell surface of 5 cell lines derived from patients with transformed MPN: UKE-1 (JAK2 mutant), HEL 92.1.7 (JAK2), SET2 (JAK2), MARIMO (CALR), MONO-MAC-6, and ELF-153 (both "triple-negative") and compared to 3 non-MPN AML lines (HL-60, MOLM-13, NOMO-1). We performed mass spectrometry in biological triplicate for each line and identified over 1,000 membrane-spanning proteins using tandem mass tag (TMT) multiplexing. Differential expression analysis identified over 70 membrane proteins enriched in MPN BP-derived cell lines and more than 120 enriched in non-MPN AML lines (Figure 1a). Gene ontology and pathway analysis suggested increased EGFR signaling in MPN, a finding published in prior reports, as well as altered lipid metabolism (Figure 1b). We then focused on C3AR1, a multi-pass G-protein coupled receptor, as a potential therapeutic target given: 1) moderate level of protein expression by proteomics, confirmed by flow cytometry (not shown); 2) enrichment in MPN-derived cells (Figure 1a); and 3) limited mRNA expression in monocytic and granulocytic cells and no significant mRNA expression in other normal tissues or endothelium (per GTEx, BloodSpot and Human Protein Atlas databases, not shown). Flow cytometry on purified CD34+ cells from marrow donors confirmed no C3AR1 expression (not shown).
Toward the second strategy, as proof-of-principle for immunotherapeutic targeting, from the literature we obtained single chain variable fragment (scFv) sequences specific for CALR (targeting a linear peptide of the N terminal domain) and C3AR1 (clone 3G7 targeting the second extracellular loop) and expressed these in a chimeric antigen receptor (CAR) backbone (IgG4 hinge with CD28 transmembrane and co-stimulatory domains). In vitro cytotoxicity assays in MARIMO cells engineered to overexpress GPI-anchored calreticulin at the cell surface showed potent activity of the anti-CALR CAR (~100% activity at 2:1 effector to tumor cell ratio in overnight co-culture) but modest activity of the anti-C3AR1 CAR (~70% lysis at a 5:1 effector to tumor cell ratio). Consistent with in vitro data, a xenotransplantation study in NSG mice showed survival benefit with anti-calreticulin CAR but lesser potency of the C3AR1 CAR (not shown).
Conclusions To our knowledge, the "surfaceome” of MPN-BP cells has not been previously profiled at the depth described here, making this dataset a unique resource to the MPN research community. These data also highlight the potential utility profiling the cell surface of MPN-BP patient samples, which may reveal additional biology or targets not captured in cell lines. While anti-calreticulin and anti-C3AR1 therapies represent promising treatment modalities for transformed MPN-BP, further work is required to increase potency in vivo. Validating surface levels of calreticulin and C3AR1 in MPN-BP patient samples, and healthy tissues, is now being pursued toward further pre-clinical development.
Disclosures
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.